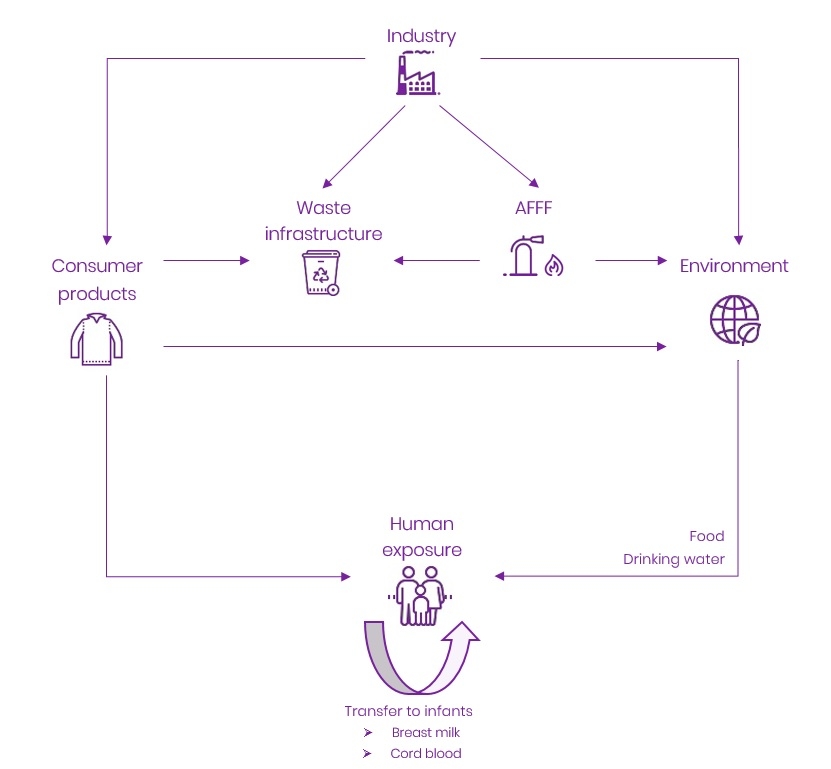
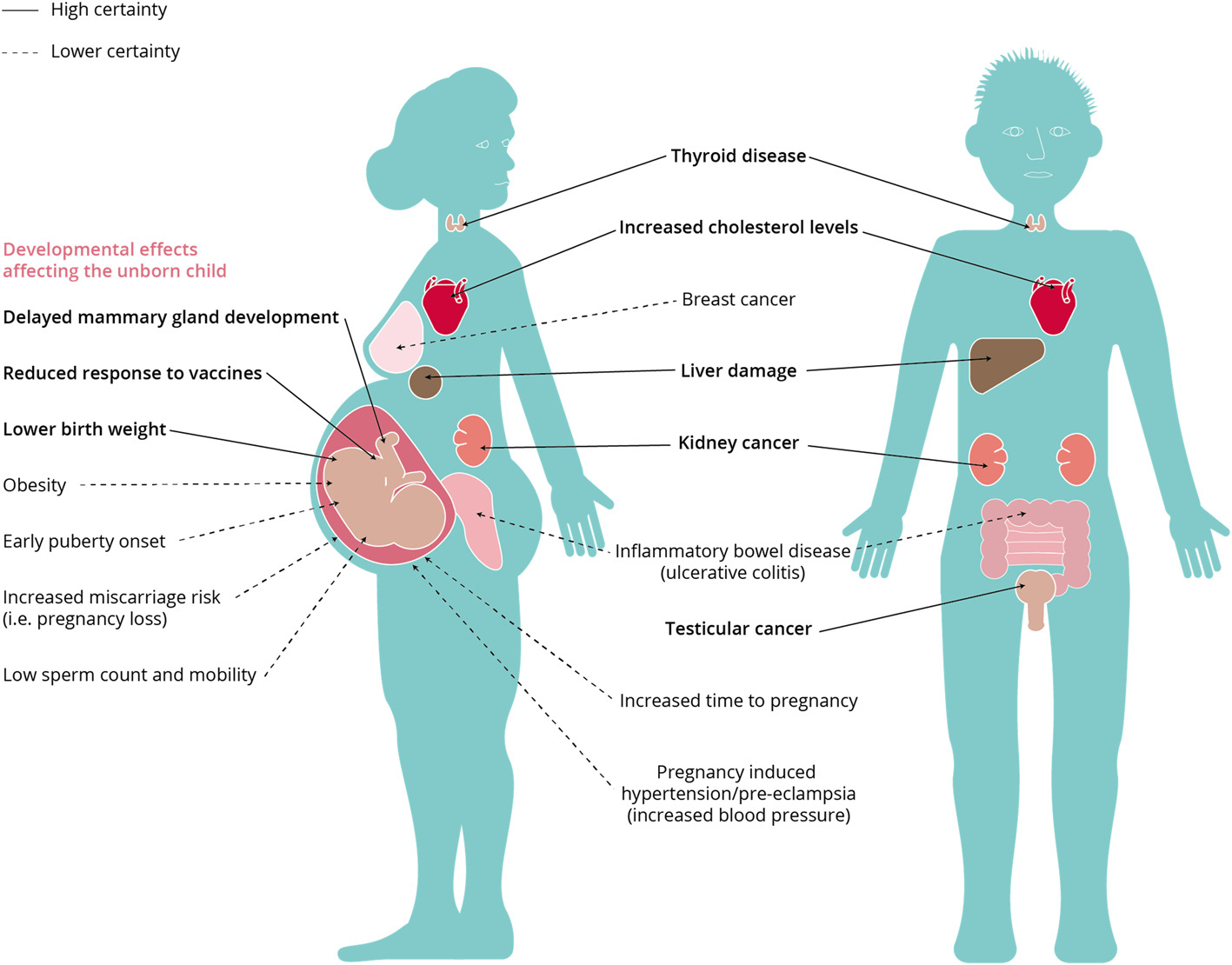
| 1 | Macmillan J. Interim Guideline on the Assessment and Management of Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) Contaminated Sites Guidelines. Perth (AU): Government of Western Australia Department of Environment and Regulation; 2016 Jan. 34 p |
| 2 | Hill PJ, Taylor M, Goswami P, Blackburn RS. Substitution of PFAS chemistry in outdoor apparel and the impact on repellency performance. Chemosphere. 2017 Aug 1;181:500-7. |
| 3 | Lee JH, Lee CK, Suh CH, Kang HS, Hong CP, Choi SN. Serum concentrations of per-and poly-fluoroalkyl substances and factors associated with exposure in the general adult population in South Korea. International Journal of Hygiene and Environmental Health. 2017 Aug 1;220(6):1046-54. |
| 4 | Bradley EL, Read WA, Castle L. Investigation into the migration potential of coating materials from cookware products. Food additives and contaminants. 2007 Mar 1;24(3):326-35. |
| 5 | Sinclair E, Kim SK, Akinleye HB, Kannan K. Quantitation of gas-phase perfluoroalkyl surfactants and fluorotelomer alcohols released from nonstick cookware and microwave popcorn bags. Environmental science & technology. 2007 Feb 15;41(4):1180-5. |
| 6 | Begley TH, White K, Honigfort P, Twaroski ML, Neches R, Walker RA. Perfluorochemicals: potential sources of and migration from food packaging. Food additives and contaminants. 2005 Oct 1;22(10):1023-31. |
| 7 | Schaider LA, Balan SA, Blum A, Andrews DQ, Strynar MJ, Dickinson ME, Lunderberg DM, Lang JR, Peaslee GF. Fluorinated compounds in US fast food packaging. Environmental science & technology letters. 2017 Mar 14;4(3):105-11. |
| 8 | Trier X, Granby K, Christensen JH. Polyfluorinated surfactants (PFS) in paper and board coatings for food packaging. Environmental Science and Pollution Research. 2011 Aug 1;18(7):1108-20. |
| 9 | Schultes L, Vestergren R, Volkova K, Westberg E, Jacobson T, Benskin JP. Per-and polyfluoroalkyl substances and fluorine mass balance in cosmetic products from the Swedish market: implications for environmental emissions and human exposure. Environmental Science: Processes & Impacts. 2018;20(12):1680-90. |
| 10 | Boronow KE, Brody JG, Schaider LA, Peaslee GF, Havas L, Cohn BA. Serum concentrations of PFASs and exposure-related behaviors in African American and non-Hispanic white women. Journal of exposure science & environmental epidemiology. 2019 Mar;29(2):206-17. |
| 11 | Xu Y, Fletcher T, Pineda D, Lindh CH, Nilsson C, Glynn A, Vogs C, Norström K, Lilja K, Jakobsson K, Li Y. Serum half-lives for short-and long-chain perfluoroalkyl acids after ceasing exposure from drinking water contaminated by firefighting foam. Environmental health perspectives. 2020 Jul 10;128(7):077004. |
| 12 | Costello MC, Lee LS. Sources, Fate, and Plant Uptake in Agricultural Systems of Per-and Polyfluoroalkyl Substances. Current Pollution Reports. 2020 Dec 15:1-21. |
| 13 | Sinclair GM, Long SM, Jones OA. What are the effects of PFAS exposure at environmentally relevant concentrations?. Chemosphere. 2020 Jun 12:127340. |
| 14 | Sunderland EM, Hu XC, Dassuncao C, Tokranov AK, Wagner CC, Allen JG. A review of the pathways of human exposure to poly-and perfluoroalkyl substances (PFASs) and present understanding of health effects. Journal of exposure science & environmental epidemiology. 2019 Mar;29(2):131-47. |
| 15 | Maestri L, Negri S, Ferrari M, Ghittori S, Fabris F, Danesino P, Imbriani M. Determination of perfluorooctanoic acid and perfluorooctanesulfonate in human tissues by liquid chromatography/single quadrupole mass spectrometry. Rapid Communications in Mass Spectrometry: An International Journal Devoted to the Rapid Dissemination of Up‐to‐the‐Minute Research in Mass Spectrometry. 2006 Sep 30;20(18):2728-34. |
| 16 | Pérez F, Nadal M, Navarro-Ortega A, Fàbrega F, Domingo JL, Barceló D, Farré M. Accumulation of perfluoroalkyl substances in human tissues. Environment international. 2013 Sep 1;59:354-62. |
| 17 | Poothong S, Thomsen C, Padilla-Sanchez JA, Papadopoulou E, Haug LS. Distribution of novel and well-known poly-and perfluoroalkyl substances (PFASs) in human serum, plasma, and whole blood. Environmental science & technology. 2017 Nov 21;51(22):13388-96. |
| 18 | Völkel W, Genzel-Boroviczény O, Demmelmair H, Gebauer C, Koletzko B, Twardella D, Raab U, Fromme H. Perfluorooctane sulphonate (PFOS) and perfluorooctanoic acid (PFOA) in human breast milk: results of a pilot study. International Journal of Hygiene and Environmental Health. 2008 Jul 15;211(3-4):440-6. |
| 19 | Johnson PI, Sutton P, Atchley DS, Koustas E, Lam J, Sen S, Robinson KA, Axelrad DA, Woodruff TJ. The Navigation Guide—evidence-based medicine meets environmental health: systematic review of human evidence for PFOA effects on fetal growth. Environmental health perspectives. 2014 Oct;122(10):1028-39. |
| 20 | Shoaff J, Papandonatos GD, Calafat AM, Chen A, Lanphear BP, Ehrlich S, Kelsey KT, Braun JM. Prenatal exposure to perfluoroalkyl substances: infant birth weight and early life growth. Environmental epidemiology (Philadelphia, Pa.). 2018 Jun;2(2). |
| 21 | Braun JM. Early-life exposure to EDCs: role in childhood obesity and neurodevelopment. Nature Reviews Endocrinology. 2017 Mar;13(3):161-73. |
| 22 | Post GB, Cohn PD, Cooper KR. Perfluorooctanoic acid (PFOA), an emerging drinking water contaminant: a critical review of recent literature. Environmental research. 2012 Jul 1;116:93-117. |
| 23 | Fenton SE, Ducatman A, Boobis A, DeWitt JC, Lau C, Ng C, Smith JS, Roberts SM. Per‐and Polyfluoroalkyl Substance Toxicity and Human Health Review: Current State of Knowledge and Strategies for Informing Future Research. Environmental Toxicology and Chemistry. 2020 Oct 5;00(00):1–25. |
| 24 | IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Some Chemicals Used as Solvents and in Polymer Manufacture. Lyon (FR): International Agency for Research on Cancer; 2017. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, No. 110. |
| 25 | Abunada Z, Alazaiza MY, Bashir MJ. An Overview of Per-and Polyfluoroalkyl Substances (PFAS) in the Environment: Source, Fate, Risk and Regulations. Water. 2020 Dec;12(12):3590. |
| 26 | Wang Z, DeWitt JC, Higgins CP, Cousins IT A Never-Ending Story of Per- and Polyfluoroalkyl Substances (PFASs)? Environmental Science & Technology 2017; 51: 2508–2518. |
| 27 | Li F, Duan J, Tian S, Ji H, Zhu Y, Wei Z, Zhao D. Short-chain per-and polyfluoroalkyl substances in aquatic systems: Occurrence, impacts and treatment. Chemical Engineering Journal. 2020 Jan 15;380:122506. |
| 28 | Tittlemier SA, Pepper K, Seymour C, Moisey J, Bronson R, Cao XL, Dabeka RW. Dietary exposure of Canadians to perfluorinated carboxylates and perfluorooctane sulfonate via consumption of meat, fish, fast foods, and food items prepared in their packaging. Journal of agricultural and food chemistry. 2007 Apr 18;55(8):3203-10. |
| 29 | Trudel D, Horowitz L, Wormuth M, Scheringer M, Cousins IT, Hungerbühler K. Estimating consumer exposure to PFOS and PFOA. Risk Analysis: An International Journal. 2008 Apr;28(2):251-69. |
| 30 | Vestergren R, Cousins IT. Tracking the pathways of human exposure to perfluorocarboxylates. Environmental science & technology. 2009 Aug 1;43(15):5565-75. |
| 31 | Haug LS, Huber S, Becher G, Thomsen C. Characterisation of human exposure pathways to perfluorinated compounds—comparing exposure estimates with biomarkers of exposure. Environment international. 2011 May 1;37(4):687-93. |
| 32 | Domingo JL, Nadal M. Per-and polyfluoroalkyl substances (PFASs) in food and human dietary intake: a review of the recent scientific literature. Journal of agricultural and food chemistry. 2017 Jan 25;65(3):533-43. |
| 33 | Domingo JL, Nadal M. Human exposure to per-and polyfluoroalkyl substances (PFAS) through drinking water: A review of the recent scientific literature. Environmental research. 2019 Oct 1;177:108648. |
| 34 | Jian JM, Guo Y, Zeng L, Liang-Ying L, Lu X, Wang F, Zeng EY. Global distribution of perfluorochemicals (PFCs) in potential human exposure source–a review. Environment international. 2017 Nov 1;108:51-62. |
| 35 | Shi Z, Zhang H, Ding L, Feng Y, Xu M, Dai J. The effect of perfluorododecanonic acid on endocrine status, sex hormones and expression of steroidogenic genes in pubertal female rats. Reproductive Toxicology. 2009 Jun 1;27(3-4):352-9. |
| 36 | Domingo JL, Jogsten IE, Eriksson U, Martorell I, Perelló G, Nadal M, van Bavel B. Human dietary exposure to perfluoroalkyl substances in Catalonia, Spain. Temporal trend. Food chemistry. 2012 Dec 1;135(3):1575-82. |
| 37 | Eriksson U, Kärrman A, Rotander A, Mikkelsen B, Dam M. Perfluoroalkyl substances (PFASs) in food and water from Faroe Islands. Environmental science and pollution research. 2013 Nov 1;20(11):7940-8. |
| 38 | Gebbink WA, Glynn A, Darnerud PO, Berger U. Perfluoroalkyl acids and their precursors in Swedish food: The relative importance of direct and indirect dietary exposure. Environmental Pollution. 2015 Mar 1;198:108-15. |
| 39 | Haug LS, Salihovic S, Jogsten IE, Thomsen C, van Bavel B, Lindström G, Becher G. Levels in food and beverages and daily intake of perfluorinated compounds in Norway. Chemosphere. 2010 Aug 1;80(10):1137-43. |
| 40 | Herzke D, Huber S, Bervoets L, D’Hollander W, Hajslova J, Pulkrabova J, Brambilla G, De Filippis SP, Klenow S, Heinemeyer G, de Voogt P. Perfluorinated alkylated substances in vegetables collected in four European countries; occurrence and human exposure estimations. Environmental science and pollution research. 2013 Nov 1;20(11):7930-9. |
| 41 | Heo JJ, Lee JW, Kim SK, Oh JE. Foodstuff analyses show that seafood and water are major perfluoroalkyl acids (PFAAs) sources to humans in Korea. Journal of hazardous materials. 2014 Aug 30;279:402-9. |
| 42 | Vestergren R, Berger U, Glynn A, Cousins IT. Dietary exposure to perfluoroalkyl acids for the Swedish population in 1999, 2005 and 2010. Environment international. 2012 Nov 15;49:120-7. |
| 43 | Zhang T, Sun HW, Wu Q, Zhang XZ, Yun SH, Kannan K. Perfluorochemicals in meat, eggs and indoor dust in China: assessment of sources and pathways of human exposure to perfluorochemicals. Environmental science & technology. 2010 May 1;44(9):3572-9. |
| 44 | Begley TH, White K, Honigfort P, Twaroski ML, Neches R, Walker RA. Perfluorochemicals: potential sources of and migration from food packaging. Food additives and contaminants. 2005 Oct 1;22(10):1023-31. |
| 45 | Schaider LA, Balan SA, Blum A, Andrews DQ, Strynar MJ, Dickinson ME, Lunderberg DM, Lang JR, Peaslee GF. Fluorinated compounds in US fast food packaging. Environmental science & technology letters. 2017 Mar 14;4(3):105-11. |
| 46 | Yuan G, Peng H, Huang C, Hu J. Ubiquitous occurrence of fluorotelomer alcohols in eco-friendly paper-made food-contact materials and their implication for human exposure. Environmental Science & Technology. 2016 Jan 19;50(2):942-50. |
| 47 | Begley TH, Hsu W, Noonan G, Diachenko G. Migration of fluorochemical paper additives from food-contact paper into foods and food simulants. Food additives and contaminants. 2008 Mar 1;25(3):384-90. |
| 48 | Susmann HP, Schaider LA, Rodgers KM, Rudel RA. Dietary Habits Related to Food Packaging and Population Exposure to PFASs. Environmental health perspectives. 2019 Oct 9;127(10):107003. |
| 49 | Cordner A, Vanessa Y, Schaider LA, Rudel RA, Richter L, Brown P. Guideline levels for PFOA and PFOS in drinking water: the role of scientific uncertainty, risk assessment decisions, and social factors. Journal of exposure science & environmental epidemiology. 2019 Mar;29(2):157-71. |
| 50 | Filipovic M, Woldegiorgis A, Norström K, Bibi M, Lindberg M, Österås AH. Historical usage of aqueous film forming foam: A case study of the widespread distribution of perfluoroalkyl acids from a military airport to groundwater, lakes, soils and fish. Chemosphere. 2015 Jun 1;129:39-45. |
| 51 | Eschauzier C, Raat KJ, Stuyfzand PJ, De Voogt P. Perfluorinated alkylated acids in groundwater and drinking water: identification, origin and mobility. Science of the total environment. 2013 Aug 1;458:477-85. |
| 52 | Szabo D, Coggan TL, Robson TC, Currell M, Clarke BO. Investigating recycled water use as a diffuse source of per-and polyfluoroalkyl substances (PFASs) to groundwater in Melbourne, Australia. Science of the Total Environment. 2018 Dec 10;644:1409-17. |
| 53 | Emmett EA, Shofer FS, Zhang H, Freeman D, Desai C, Shaw LM. Community exposure to perfluorooctanoate: relationships between serum concentrations and exposure sources. Journal of occupational and environmental medicine/American College of Occupational and Environmental Medicine. 2006 Aug;48(8):759-70. |
| 54 | Seals R, Bartell SM, Steenland K. Accumulation and clearance of perfluorooctanoic acid (PFOA) in current and former residents of an exposed community. Environmental health perspectives. 2011 Jan;119(1):119-24. |
| 55 | Thompson J, Eaglesham G, Mueller J. Concentrations of PFOS, PFOA and other perfluorinated alkyl acids in Australian drinking water. Chemosphere. 2011 May 1;83(10):1320-5. |
| 56 | Akhbarizadeh R, Dobaradaran S, Schmidt TC, Nabipour I, Spitz J. Worldwide bottled water occurrence of emerging contaminants: A review of the recent scientific literature. Journal of Hazardous Materials. 2020 Jun 15;392:122271. |
| 57 | Llorca M, Farré M, Picó Y, Müller J, Knepper TP, Barceló D. Analysis of perfluoroalkyl substances in waters from Germany and Spain. Science of the total environment. 2012 Aug 1;431:139-50. |
| 58 | Schwanz TG, Llorca M, Farré M, Barceló D. Perfluoroalkyl substances assessment in drinking waters from Brazil, France and Spain. Science of the total environment. 2016 Jan 1;539:143-52. |
| 59 | Glüge J, Scheringer M, Cousins IT, DeWitt JC, Goldenman G, Herzke D, Lohmann R, Ng CA, Trier X, Wang Z. An overview of the uses of per-and polyfluoroalkyl substances (PFAS). Environmental Science: Processes & Impacts. 2020;22(12):2345-73. |
| 60 | Kotthoff M, Müller J, Jürling H, Schlummer M, Fiedler D. Perfluoroalkyl and polyfluoroalkyl substances in consumer products. Environmental Science and Pollution Research. 2015 Oct 1;22(19):14546-59. |
| 61 | Haug LS, Huber S, Schlabach M, Becher G, Thomsen C. Investigation on per-and polyfluorinated compounds in paired samples of house dust and indoor air from Norwegian homes. Environmental science & technology. 2011 Oct 1;45(19):7991-8. |
| 62 | Langer V, Dreyer A, Ebinghaus R. Polyfluorinated compounds in residential and nonresidential indoor air. Environmental science & technology. 2010 Nov 1;44(21):8075-81. |
| 63 | Yao Y, Zhao Y, Sun H, Chang S, Zhu L, Alder AC, Kannan K. Per-and polyfluoroalkyl substances (PFASs) in indoor air and dust from homes and various microenvironments in China: implications for human exposure. Environmental science & technology. 2018 Feb 8;52(5):3156-66. |
| 64 | Ding N, Harlow SD, Randolph JF, Loch-Caruso R, Park SK. Perfluoroalkyl and polyfluoroalkyl substances (PFAS) and their effects on the ovary. Human Reproduction Update. 2020 May 29;26(5):724–52. |
| 65 | Kashino I, Sasaki S, Okada E, Matsuura H, Goudarzi H, Miyashita C, Okada E, Ito YM, Araki A, Kishi R. Prenatal exposure to 11 perfluoroalkyl substances and fetal growth: A large-scale, prospective birth cohort study. Environment International. 2020 Mar 1;136:105355. |
| 66 | Lee YJ, Kim MK, Bae J, Yang JH. Concentrations of perfluoroalkyl compounds in maternal and umbilical cord sera and birth outcomes in Korea. Chemosphere. 2013 Feb 1;90(5):1603-9. |
| 67 | Pan Y, Zhu Y, Zheng T, Cui Q, Buka SL, Zhang B, Guo Y, Xia W, Yeung LW, Li Y, Zhou A. Novel chlorinated polyfluorinated ether sulfonates and legacy per-/polyfluoroalkyl substances: placental transfer and relationship with serum albumin and glomerular filtration rate. Environmental science & technology. 2017 Jan 3;51(1):634-44. |
| 68 | Genuis SJ, Birkholz D, Ralitsch M, Thibault N. Human detoxification of perfluorinated compounds. Public health. 2010 Jul 1;124(7):367-75. |
| 69 | Dong Z, Wang H, Yu YY, Li YB, Naidu R, Liu Y. Using 2003–2014 US NHANES data to determine the associations between per-and polyfluoroalkyl substances and cholesterol: Trend and implications. Ecotoxicology and environmental safety. 2019 May 30;173:461-8. |
| 70 | Li Y, Barregard L, Xu Y, Scott K, Pineda D, Lindh CH, Jakobsson K, Fletcher T. Associations between perfluoroalkyl substances and serum lipids in a Swedish adult population with contaminated drinking water. Environmental Health. 2020 Dec;19(1):1-1. |
| 71 | Lin PI, Cardenas A, Hauser R, Gold DR, Kleinman KP, Hivert MF, Fleisch AF, Calafat AM, Webster TF, Horton ES, Oken E. Per-and polyfluoroalkyl substances and blood lipid levels in pre-diabetic adults—longitudinal analysis of the diabetes prevention program outcomes study. Environment international. 2019 Aug 1;129:343-53. |
| 72 | Steenland K, Tinker S, Frisbee S, Ducatman A, Vaccarino V. Association of perfluorooctanoic acid and perfluorooctane sulfonate with serum lipids among adults living near a chemical plant. American journal of epidemiology. 2009 Nov 15;170(10):1268-78. |
| 73 | Winquist A, Steenland K. Modeled PFOA exposure and coronary artery disease, hypertension, and high cholesterol in community and worker cohorts. Environmental health perspectives. 2014 Dec;122(12):1299-305. |
| 74 | Zeng XW, Qian Z, Emo B, Vaughn M, Bao J, Qin XD, Zhu Y, Li J, Lee YL, Dong GH. Association of polyfluoroalkyl chemical exposure with serum lipids in children. Science of the Total Environment. 2015 Apr 15;512:364-70. |
| 75 | Alderete TL, Jin R, Walker DI, Valvi D, Chen Z, Jones DP, Peng C, Gilliland FD, Berhane K, Conti DV, Goran MI. Perfluoroalkyl substances, metabolomic profiling, and alterations in glucose homeostasis among overweight and obese Hispanic children: A proof-of-concept analysis. Environment international. 2019 May 1;126:445-53. |
| 76 | Sun Q, Zong G, Valvi D, Nielsen F, Coull B, Grandjean P. Plasma concentrations of perfluoroalkyl substances and risk of type 2 diabetes: a prospective investigation among US women. Environmental health perspectives. 2018 Mar 1;126(3):037001. |
| 77 | Cardenas A, Gold DR, Hauser R, Kleinman KP, Hivert MF, Calafat AM, Ye X, Webster TF, Horton ES, Oken E. Plasma concentrations of per-and polyfluoroalkyl substances at baseline and associations with glycemic indicators and diabetes incidence among high-risk adults in the Diabetes Prevention Program Trial. Environmental health perspectives. 2017 Oct 2;125(10):107001. |
| 78 | Karnes C, Winquist A, Steenland K. Incidence of type II diabetes in a cohort with substantial exposure to perfluorooctanoic acid. Environmental research. 2014 Jan 1;128:78-83. |
| 79 | Donat-Vargas C, Bergdahl IA, Tornevi A, Wennberg M, Sommar J, Kiviranta H, Koponen J, Rolandsson O, Åkesson A. Perfluoroalkyl substances and risk of type II diabetes: A prospective nested case-control study. Environment international. 2019 Feb 1;123:390-8. |
| 80 | Grandjean P, Andersen EW, Budtz-Jørgensen E, Nielsen F, Mølbak K, Weihe P, Heilmann C. Serum vaccine antibody concentrations in children exposed to perfluorinated compounds. Jama. 2012 Jan 25;307(4):391-7. |
| 81 | Grandjean P, Heilmann C, Weihe P, Nielsen F, Mogensen UB, Budtz-Jørgensen E. Serum vaccine antibody concentrations in adolescents exposed to perfluorinated compounds. Environmental Health Perspectives. 2017 Jul 26;125(7):077018. |
| 82 | Granum B, Haug LS, Namork E, Stølevik SB, Thomsen C, Aaberge IS, van Loveren H, Løvik M, Nygaard UC. Pre-natal exposure to perfluoroalkyl substances may be associated with altered vaccine antibody levels and immune-related health outcomes in early childhood. Journal of immunotoxicology. 2013 Oct 1;10(4):373-9. |
| 83 | Goudarzi H, Miyashita C, Okada E, Kashino I, Chen CJ, Ito S, Araki A, Kobayashi S, Matsuura H, Kishi R. Prenatal exposure to perfluoroalkyl acids and prevalence of infectious diseases up to 4 years of age. Environment international. 2017 Jul 1;104:132-8. |
| 84 | Impinen A, Longnecker MP, Nygaard UC, London SJ, Ferguson KK, Haug LS, Granum B. Maternal levels of perfluoroalkyl substances (PFASs) during pregnancy and childhood allergy and asthma related outcomes and infections in the Norwegian mother and child (MoBa) cohort. Environment international. 2019 Mar 1;124:462-72. |
| 85 | Impinen A, Nygaard UC, Carlsen KL, Mowinckel P, Carlsen KH, Haug LS, Granum B. Prenatal exposure to perfluoralkyl substances (PFASs) associated with respiratory tract infections but not allergy-and asthma-related health outcomes in childhood. Environmental research. 2018 Jan 1;160:518-23. |
| 86 | Looker C, Luster MI, Calafat AM, Johnson VJ, Burleson GR, Burleson FG, Fletcher T. Influenza vaccine response in adults exposed to perfluorooctanoate and perfluorooctanesulfonate. toxicological sciences. 2014 Mar 1;138(1):76-88. |
| 87 | Qin XD, Qian ZM, Dharmage SC, Perret J, Geiger SD, Rigdon SE, Howard S, Zeng XW, Hu LW, Yang BY, Zhou Y. Association of perfluoroalkyl substances exposure with impaired lung function in children. Environmental research. 2017 May 1;155:15-21. |
| 88 | Timmermann CA, Budtz-Jørgensen E, Jensen TK, Osuna CE, Petersen MS, Steuerwald U, Nielsen F, Poulsen LK, Weihe P, Grandjean P. Association between perfluoroalkyl substance exposure and asthma and allergic disease in children as modified by MMR vaccination. Journal of immunotoxicology. 2017 Jan 1;14(1):39-49. |
| 89 | Kvalem HE, Nygaard UC, Carlsen KL, Carlsen KH, Haug LS, Granum B. Perfluoroalkyl substances, airways infections, allergy and asthma related health outcomes–implications of gender, exposure period and study design. Environment international. 2020 Jan 1;134:105259. |
| 90 | Steenland K, Zhao L, Winquist A, Parks C. Ulcerative colitis and perfluorooctanoic acid (PFOA) in a highly exposed population of community residents and workers in the mid-Ohio valley. Environmental health perspectives. 2013 Aug;121(8):900-5. |
| 91 | Attanasio R. Sex differences in the association between perfluoroalkyl acids and liver function in US adolescents: Analyses of NHANES 2013–2016. Environmental Pollution. 2019 Nov 1;254:113061. |
| 92 | Darrow LA, Groth AC, Winquist A, Shin HM, Bartell SM, Steenland K. Modeled perfluorooctanoic acid (PFOA) exposure and liver function in a mid-Ohio valley community. Environmental health perspectives. 2016 Aug;124(8):1227-33. |
| 93 | Gallo V, Leonardi G, Genser B, Lopez-Espinosa MJ, Frisbee SJ, Karlsson L, Ducatman AM, Fletcher T. Serum perfluorooctanoate (PFOA) and perfluorooctane sulfonate (PFOS) concentrations and liver function biomarkers in a population with elevated PFOA exposure. Environmental health perspectives. 2012 May;120(5):655-60. |
| 94 | Gleason JA, Post GB, Fagliano JA. Associations of perfluorinated chemical serum concentrations and biomarkers of liver function and uric acid in the US population (NHANES), 2007–2010. Environmental research. 2015 Jan 1;136:8-14. |
| 95 | Nian M, Li QQ, Bloom M, Qian ZM, Syberg KM, Vaughn MG, Wang SQ, Wei Q, Zeeshan M, Gurram N, Chu C. Liver function biomarkers disorder is associated with exposure to perfluoroalkyl acids in adults: Isomers of C8 Health Project in China. Environmental research. 2019 May 1;172:81-8. |
| 96 | Sakr CJ, Kreckmann KH, Green JW, Gillies PJ, Reynolds JL, Leonard RC. Cross-sectional study of lipids and liver enzymes related to a serum biomarker of exposure (ammonium perfluorooctanoate or APFO) as part of a general health survey in a cohort of occupationally exposed workers. Journal of occupational and environmental medicine. 2007 Oct 1;49(10):1086-96. |
| 97 | Sakr CJ, Leonard RC, Kreckmann KH, Slade MD, Cullen MR. Longitudinal study of serum lipids and liver enzymes in workers with occupational exposure to ammonium perfluorooctanoate. Journal of occupational and environmental medicine. 2007 Aug 1;49(8):872-9. |
| 98 | Yamaguchi M, Arisawa K, Uemura H, Katsuura‐Kamano S, Takami H, Sawachika F, Nakamoto M, Juta T, Toda E, Mori K, Hasegawa M. Consumption of seafood, serum liver enzymes, and blood levels of PFOS and PFOA in the Japanese population. Journal of occupational health. 2013 May;55(3):184-94. |
| 99 | Geiger SD, Xiao J, Shankar A. Positive association between perfluoroalkyl chemicals and hyperuricemia in children. American journal of epidemiology. 2013 Jun 1;177(11):1255-62. |
| 100 | Obermayr RP, Temml C, Gutjahr G, Knechtelsdorfer M, Oberbauer R, Klauser-Braun R. Elevated uric acid increases the risk for kidney disease. Journal of the American Society of Nephrology. 2008 Dec 1;19(12):2407-13. |
| 101 | Qin XD, Qian Z, Vaughn MG, Huang J, Ward P, Zeng XW, Zhou Y, Zhu Y, Yuan P, Li M, Bai Z. Positive associations of serum perfluoroalkyl substances with uric acid and hyperuricemia in children from Taiwan. Environmental pollution. 2016 May 1;212:519-24. |
| 102 | Steenland K, Tinker S, Shankar A, Ducatman A. Association of perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) with uric acid among adults with elevated community exposure to PFOA. Environmental health perspectives. 2010 Feb;118(2):229-33. |
| 103 | Zeng XW, Lodge CJ, Dharmage SC, Bloom MS, Yu Y, Yang M, Chu C, Li QQ, Hu LW, Liu KK, Yang BY. Isomers of per-and polyfluoroalkyl substances and uric acid in adults: Isomers of C8 Health Project in China. Environment international. 2019 Dec 1;133:105160. |
| 104 | Buckley N, Sim M, Douglas K, Håkansson H. Expert Health Panel for Per- and Poly-Fluoroalkyl Substances (PFAS). Department of Health AG; 2018 March:446p. |
| 105 | Kato S, Itoh S, Yuasa M, Baba T, Miyashita C, Sasaki S, et al. Association of perfluorinated chemical exposure in utero with maternal and infant thyroid hormone levels in the Sapporo cohort of Hokkaido Study on the Environment and Children's Health. Environ Health Prev Med. 2016;21(5):334–44. |
| 106 | Kim S, Choi K, Ji K, Seo J, Kho Y, Park J, et al. Trans-placental transfer of thirteen perfluorinated compounds and relations with fetal thyroid hormones. Environ Sci Technol. 2011;45(17):7465–72. |
| 107 | Inoue K, Ritz B, Andersen SL, Ramlau-Hansen CH, Høyer BB, Bech BH, Henriksen TB, Bonefeld-Jørgensen EC, Olsen J, Liew Z. Perfluoroalkyl substances and maternal thyroid hormones in early pregnancy; findings in the Danish National Birth Cohort. Environmental health perspectives. 2019 Nov 12;127(11):117002. |
| 108 | Coperchini F, Awwad O, Rotondi M, Santini FE, Imbriani M, Chiovato L. Thyroid disruption by perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA). Journal of endocrinological investigation. 2017 Feb 1;40(2):105-21. |
| 109 | Rylander L, Lindh CH, Hansson SR, Broberg K, Källén K. Per-and Polyfluoroalkyl Substances in Early Pregnancy and Risk for Preeclampsia: A Case-Control Study in Southern Sweden. Toxics. 2020 Jun;8(2):43. |
| 110 | Savitz DA, Stein CR, Bartell SM, Elston B, Gong J, Shin HM, Wellenius GA. Perfluorooctanoic acid exposure and pregnancy outcome in a highly exposed community. Epidemiology (Cambridge, Mass.). 2012 May;23(3):386. |
| 111 | Stein CR, Savitz DA, Dougan M. Serum levels of perfluorooctanoic acid and perfluorooctane sulfonate and pregnancy outcome. American journal of epidemiology. 2009 Oct 1;170(7):837-46. |
| 112 | Bach CC, Bech BH, Nohr EA, Olsen J, Matthiesen NB, Bossi R, Uldbjerg N, Bonefeld-Jørgensen EC, Henriksen TB. Serum perfluoroalkyl acids and time to pregnancy in nulliparous women. Environmental research. 2015 Oct 1;142:535-41. |
| 113 | Starling AP, Adgate JL, Hamman RF, Kechris K, Calafat AM, Ye X, Dabelea D. Perfluoroalkyl substances during pregnancy and offspring weight and adiposity at birth: examining mediation by maternal fasting glucose in the healthy start study. Environmental health perspectives. 2017 Jun 26;125(6):067016. |
| 114 | Wikström S, Lin PI, Lindh CH, Shu H, Bornehag CG. Maternal serum levels of perfluoroalkyl substances in early pregnancy and offspring birth weight. Pediatric Research. 2020;87(6):1093. |
| 115 | Xiao C, Grandjean P, Valvi D, Nielsen F, Jensen TK, Weihe P, Oulhote Y. Associations of exposure to perfluoroalkyl substances with thyroid hormone concentrations and birth size. The Journal of Clinical Endocrinology & Metabolism. 2020 Mar;105(3):735-45. |
| 116 | Mora AM, Oken E, Rifas-Shiman SL, Webster TF, Gillman MW, Calafat AM, Ye X, Sagiv SK. Prenatal exposure to perfluoroalkyl substances and adiposity in early and mid-childhood. Environmental health perspectives. 2017 Mar;125(3):467-73. |
| 117 | Chen Q, Zhang X, Zhao Y, Lu W, Wu J, Zhao S, Zhang J, Huang L. Prenatal exposure to perfluorobutanesulfonic acid and childhood adiposity: A prospective birth cohort study in Shanghai, China. Chemosphere. 2019 Jul 1;226:17-23. |
| 118 | Fleisch AF, Rifas-Shiman SL, Mora AM, Calafat AM, Ye X, Luttmann-Gibson H, Gillman MW, Oken E, Sagiv SK. Early-life exposure to perfluoroalkyl substances and childhood metabolic function. Environmental health perspectives. 2017 Mar;125(3):481-7. |
| 119 | Gardener H, Sun Q, Grandjean P. PFAS concentration during pregnancy in relation to cardiometabolic health and birth outcomes. Environmental Research. 2020 Oct 8;192:110287. |
| 120 | Lopez-Espinosa MJ, Mondal D, Armstrong BG, Eskenazi B, Fletcher T. Perfluoroalkyl substances, sex hormones, and insulin-like growth factor-1 at 6–9 years of age: a cross-sectional analysis within the C8 Health Project. Environmental health perspectives. 2016 Aug;124(8):1269-75. |
| 121 | Nelson JW, Hatch EE, Webster TF. Exposure to polyfluoroalkyl chemicals and cholesterol, body weight, and insulin resistance in the general US population. Environmental health perspectives. 2010 Feb;118(2):197-202. |
| 122 | Rappazzo KM, Coffman E, Hines EP. Exposure to perfluorinated alkyl substances and health outcomes in children: a systematic review of the epidemiologic literature. International journal of environmental research and public health. 2017 Jul;14(7):691. |
| 123 | Gump BB, Wu Q, Dumas AK, Kannan K. Perfluorochemical (PFC) exposure in children: associations with impaired response inhibition. Environmental science & technology. 2011 Oct 1;45(19):8151-9. |
| 124 | Hoffman K, Webster TF, Weisskopf MG, Weinberg J, Vieira VM. Exposure to polyfluoroalkyl chemicals and attention deficit/hyperactivity disorder in US children 12–15 years of age. Environmental health perspectives. 2010 Dec;118(12):1762-7. |
| 125 | Høyer BB, Ramlau-Hansen CH, Obel C, Pedersen HS, Hernik A, Ogniev V, Jönsson BA, Lindh CH, Rylander L, Rignell-Hydbom A, Bonde JP. Pregnancy serum concentrations of perfluorinated alkyl substances and offspring behaviour and motor development at age 5–9 years–a prospective study. Environmental Health. 2015 Dec 1;14(1):2. |
| 126 | Stein CR, Savitz DA. Serum perfluorinated compound concentration and attention deficit/hyperactivity disorder in children 5–18 years of age. Environmental health perspectives. 2011 Oct;119(10):1466-71. |
| 127 | Zhou W, Zhang L, Tong C, Fang F, Zhao S, Tian Y, Tao Y, Zhang J, Shanghai Birth Cohort Study. Plasma perfluoroalkyl and polyfluoroalkyl substances concentration and menstrual cycle characteristics in preconception women. Environmental health perspectives. 2017 Jun 22;125(6):067012. |
| 128 | Taylor KW, Hoffman K, Thayer KA, Daniels JL. Polyfluoroalkyl chemicals and menopause among women 20–65 years of age (NHANES). Environmental health perspectives. 2014 Feb;122(2):145-50. |
| 129 | Zhang S, Tan R, Pan R, Xiong J, Tian Y, Wu J, Chen L. Association of perfluoroalkyl and polyfluoroalkyl substances with premature ovarian insufficiency in Chinese women. The Journal of Clinical Endocrinology & Metabolism. 2018 Jul;103(7):2543-51. |
| 130 | Barrett ES, Chen C, Thurston SW, Haug LS, Sabaredzovic A, Fjeldheim FN, Frydenberg H, Lipson SF, Ellison PT, Thune I. Perfluoroalkyl substances and ovarian hormone concentrations in naturally cycling women. Fertility and sterility. 2015 May 1;103(5):1261-70. |
| 131 | Ding N, Harlow SD, Randolph JF, Calafat AM, Mukherjee B, Batterman S, Gold EB, Park SK. Associations of Perfluoroalkyl Substances with Incident Natural Menopause: the Study of Women’s Health Across the Nation. The Journal of Clinical Endocrinology & Metabolism. 2020 Jun 3;105(9):e3169-82. |
| 132 | Heffernan AL, Cunningham TK, Drage DS, Aylward LL, Thompson K, Vijayasarathy S, Mueller JF, Atkin SL, Sathyapalan T. Perfluorinated alkyl acids in the serum and follicular fluid of UK women with and without polycystic ovarian syndrome undergoing fertility treatment and associations with hormonal and metabolic parameters. International Journal of Hygiene and Environmental Health. 2018 Aug 1;221(7):1068-75. |
| 133 | Vagi SJ, Azziz-Baumgartner E, Sjödin A, Calafat AM, Dumesic D, Gonzalez L, Kato K, Silva MJ, Ye X, Azziz R. Exploring the potential association between brominated diphenyl ethers, polychlorinated biphenyls, organochlorine pesticides, perfluorinated compounds, phthalates, and bisphenol a in polycystic ovary syndrome: a case–control study. BMC endocrine disorders. 2014 Dec 1;14(1):86. |
| 134 | Campbell S, Raza M, Pollack AZ. Perfluoroalkyl substances and endometriosis in US women in NHANES 2003–2006. Reproductive toxicology. 2016 Oct 1;65:230-5. |
| 135 | Wang B, Zhang R, Jin F, Lou H, Mao Y, Zhu W, Zhou W, Zhang P, Zhang J. Perfluoroalkyl substances and endometriosis-related infertility in Chinese women. Environment international. 2017 May 1;102:207-12. |
| 136 | Fei C, McLaughlin JK, Lipworth L, Olsen J. Maternal levels of perfluorinated chemicals and subfecundity. Human reproduction. 2009 May 1;24(5):1200-5. |
| 137 | Jørgensen KT, Specht IO, Lenters V, Bach CC, Rylander L, Jönsson BA, Lindh CH, Giwercman A, Heederik D, Toft G, Bonde JP. Perfluoroalkyl substances and time to pregnancy in couples from Greenland, Poland and Ukraine. Environmental health. 2014 Dec 1;13(1):116. |
| 138 | Vélez MP, Arbuckle TE, Fraser WD. Maternal exposure to perfluorinated chemicals and reduced fecundity: the MIREC study. Hum Reprod. 2015 Mar;30(3):701-9. |
| 139 | Whitworth KW, Haug LS, Baird DD, Becher G, Hoppin JA, Skjaerven R, Thomsen C, Eggesbo M, Travlos G, Wilson R, Longnecker MP. Perfluorinated compounds and subfecundity in pregnant women. Epidemiology (Cambridge, Mass.). 2012 Mar;23(2):257. |
| 140 | Bach CC, Bech BH, Nohr EA, Olsen J, Matthiesen NB, Bossi R, Uldbjerg N, Bonefeld-Jørgensen EC, Henriksen TB. Serum perfluoroalkyl acids and time to pregnancy in nulliparous women. Environmental research. 2015 Oct 1;142:535-41. |
| 141 | Louis GM, Sundaram R, Schisterman EF, Sweeney AM, Lynch CD, Gore-Langton RE, Maisog J, Kim S, Chen Z, Barr DB. Persistent environmental pollutants and couple fecundity: the LIFE study. Environmental health perspectives. 2013 Feb;121(2):231-6. |
| 142 | Vestergaard S, Nielsen F, Andersson AM, Hjøllund NH, Grandjean P, Andersen HR, Jensen TK. Association between perfluorinated compounds and time to pregnancy in a prospective cohort of Danish couples attempting to conceive. Human reproduction. 2012 Mar 1;27(3):873-80. |
| 143 | Joensen UN, Bossi R, Leffers H, Jensen AA, Skakkebæk NE, Jørgensen N. Do perfluoroalkyl compounds impair human semen quality?. Environmental health perspectives. 2009 Jun;117(6):923-7. |
| 144 | Joensen UN, Veyrand B, Antignac JP, Blomberg Jensen M, Petersen JH, Marchand P, Skakkebaek NE, Andersson AM, Le Bizec B, Jørgensen N. PFOS (perfluorooctanesulfonate) in serum is negatively associated with testosterone levels, but not with semen quality, in healthy men. Human reproduction. 2013 Mar 1;28(3):599-608. |
| 145 | Lopez-Espinosa MJ, Mondal D, Armstrong BG, Eskenazi B, Fletcher T. Perfluoroalkyl substances, sex hormones, and insulin-like growth factor-1 at 6–9 years of age: a cross-sectional analysis within the C8 Health Project. Environmental health perspectives. 2016 Aug;124(8):1269-75. |
| 146 | Raymer JH, Michael LC, Studabaker WB, Olsen GW, Sloan CS, Wilcosky T, Walmer DK. Concentrations of perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) and their associations with human semen quality measurements. Reproductive toxicology. 2012 Jul 1;33(4):419-27. |
| 147 | Zhou Y, Hu LW, Qian ZM, Chang JJ, King C, Paul G, Lin S, Chen PC, Lee YL, Dong GH. Association of perfluoroalkyl substances exposure with reproductive hormone levels in adolescents: By sex status. Environment international. 2016 Sep 1;94:189-95. |
| 148 | Louis GM, Chen Z, Schisterman EF, Kim S, Sweeney AM, Sundaram R, Lynch CD, Gore-Langton RE, Barr DB. Perfluorochemicals and human semen quality: the LIFE study. Environmental health perspectives. 2015 Jan;123(1):57-63. |
| 149 | Pan Y, Cui Q, Wang J, Sheng N, Jing J, Yao B, Dai J. Profiles of Emerging and Legacy Per-/Polyfluoroalkyl Substances in Matched Serum and Semen Samples: New Implications for Human Semen Quality. Environmental health perspectives. 2019 Dec 16;127(12):127005. |
| 150 | Environmental Protection Agency. Drinking Water Health Advisory for Perfluorooctanoic Acid (PFOA). Washington, DC, USA, US Environmental Protection Agency Office of Water (4304T) Health and Ecological Criteria Division; 2016. EPA Document Number: 822-R-16-005. |
| 151 | Environmental Protection Agency. Drinking Water Health Advisory for Perfluorooctane Sulfonate (PFOS). Washington, DC, USA, US Environmental Protection Agency Office of Water (4304T) Health and Ecological Criteria Division; 2016. EPA Document Number: 822-R-16-004 |
| 152 | Environmental Protection Agency. Human Health Toxicity Values for Hexafluoropropylene Oxide (HFPO) Dimer Acid and Its Ammonium Salt (CASRN 13252-13-6 and CASRN 62037-80-3) Also Known as “GenX Chemicals”. Washington, DC, USA, US Environmental Protection Agency Office of Water (4304T) Health and Ecological Criteria Division; 2018. EPA Document Number: EPA Document Number: 823-P-18-001. |
| 153 | Barry V, Winquist A, Steenland K. Perfluorooctanoic acid (PFOA) exposures and incident cancers among adults living near a chemical plant. Environmental health perspectives. 2013;121(11-12):1313-8. |
| 154 | Vieira VM, Hoffman K, Shin HM, Weinberg JM, Webster TF, Fletcher T. Perfluorooctanoic acid exposure and cancer outcomes in a contaminated community: a geographic analysis. Environ Health Perspect. 2013 Mar;121(3):318-23. |
| 155 | Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological profile for Perfluoroalkyls. (Draft for Public Comment). Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service. 2018; 852 p. |
| 156 | Kemp MJ, Boyd CA. PFAS Toxicology–The Science Behind the Variation in Drinking Water Standards. Journal of the New England Water Works Association. 2019 Dec 1;133(4):243-55. |
| 157 | Genuis SJ, Curtis L, Birkholz D. Gastrointestinal elimination of perfluorinated compounds using cholestyramine and Chlorella pyrenoidosa. International Scholarly Research Notices. 2013;2013. |
| 158 | Behr AC, Plinsch C, Braeuning A, Buhrke T. Activation of human nuclear receptors by perfluoroalkylated substances (PFAS). Toxicology in Vitro. 2020 Feb 1;62:104700. |
| 159 | White SS, Fenton SE, Hines EP. Endocrine disrupting properties of perfluorooctanoic acid. The Journal of steroid biochemistry and molecular biology. 2011 Oct 1;127(1-2):16-26. |
| 160 | Huang Q, Chen Q. Mediating roles of PPARs in the effects of environmental chemicals on sex steroids. Ppar Research. 2017 Oct;2017. |
| 161 | Rosen MB, Lee JS, Ren H, Vallanat B, Liu J, Waalkes MP, Abbott BD, Lau C, Corton JC. Toxicogenomic dissection of the perfluorooctanoic acid transcript profile in mouse liver: evidence for the involvement of nuclear receptors PPARα and CAR. Toxicological Sciences. 2008 May 1;103(1):46-56. |
| 162 | Rosen MB, Das KP, Rooney J, Abbott B, Lau C, Corton JC. PPARα-independent transcriptional targets of perfluoroalkyl acids revealed by transcript profiling. Toxicology. 2017 Jul 15;387:95-107. |
| 163 | Wolf CJ, Schmid JE, Lau C, Abbott BD. Activation of mouse and human peroxisome proliferator-activated receptor-alpha (PPARα) by perfluoroalkyl acids (PFAAs): further investigation of C4–C12 compounds. Reproductive toxicology. 2012 Jul 1;33(4):546-51. |
| 164 | Zhang L, Ren XM, Wan B, Guo LH. Structure-dependent binding and activation of perfluorinated compounds on human peroxisome proliferator-activated receptor γ. Toxicology and applied pharmacology. 2014 Sep 15;279(3):275-83. |
| 165 | Behr AC, Kwiatkowski A, Ståhlman M, Schmidt FF, Luckert C, Braeuning A, Buhrke T. Impairment of bile acid metabolism by perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) in human HepaRG hepatoma cells. Archives of Toxicology. 2020 Apr 6:1-4. |
| 166 | Bijland S, Rensen PC, Pieterman EJ, Maas AC, van der Hoorn JW, van Erk MJ, Havekes LM, Willems van Dijk K, Chang SC, Ehresman DJ, Butenhoff JL. Perfluoroalkyl sulfonates cause alkyl chain length–dependent hepatic steatosis and hypolipidemia mainly by impairing lipoprotein production in APOE* 3-Leiden CETP mice. Toxicological Sciences. 2011 Sep 1;123(1):290-303. |
| 167 | Bjork JA, Butenhoff JL, Wallace KB. Multiplicity of nuclear receptor activation by PFOA and PFOS in primary human and rodent hepatocytes. Toxicology. 2011 Oct 9;288(1-3):8-17. |
| 168 | Das KP, Wood CR, Lin MT, Starkov AA, Lau C, Wallace KB, Corton JC, Abbott BD. Perfluoroalkyl acids-induced liver steatosis: Effects on genes controlling lipid homeostasis. Toxicology. 2017 Mar 1;378:37-52. |
| 169 | Guruge KS, Yeung LW, Yamanaka N, Miyazaki S, Lam PK, Giesy JP, Jones PD, Yamashita N. Gene expression profiles in rat liver treated with perfluorooctanoic acid (PFOA). Toxicological Sciences. 2006 Jan 1;89(1):93-107. |
| 170 | Salihovic S, Fall T, Ganna A, Broeckling CD, Prenni JE, Hyötyläinen T, Kärrman A, Lind PM, Ingelsson E, Lind L. Identification of metabolic profiles associated with human exposure to perfluoroalkyl substances. Journal of exposure science & environmental epidemiology. 2019 Mar;29(2):196-205. |
| 171 | Schlezinger J, Puckett H, Oliver J, Nielsen G, Heiger-Bernays W, Webster T. Perfluorooctanoic acid activates multiple nuclear receptor pathways and skews expression of genes regulating cholesterol homeostasis in liver of humanized PPARα mice fed an American diet. 2020 Oct 15; 405:115204. |
| 172 | Zhang H, He J, Li N, Gao N, Du Q, Chen B, Chen F, Shan X, Ding Y, Zhu W, Wu Y. Lipid accumulation responses in the liver of Rana nigromaculata induced by perfluorooctanoic acid (PFOA). Ecotoxicology and environmental safety. 2019 Jan 15;167:29-35. |
| 173 | Berg V, Nøst TH, Hansen S, Elverland A, Veyhe AS, Jorde R, Odland JØ, Sandanger TM. Assessing the relationship between perfluoroalkyl substances, thyroid hormones and binding proteins in pregnant women; a longitudinal mixed effects approach. Environment international. 2015 Apr 1;77:63-9. |
| 174 | Song M, Kim YJ, Park YK, Ryu JC. Changes in thyroid peroxidase activity in response to various chemicals. Journal of environmental monitoring. 2012;14(8):2121-6. |
| 175 | Benninghoff AD, Bisson WH, Koch DC, Ehresman DJ, Kolluri SK, Williams DE. Estrogen-like activity of perfluoroalkyl acids in vivo and interaction with human and rainbow trout estrogen receptors in vitro. Toxicological sciences. 2011 Mar 1;120(1):42-58. |
| 176 | Hemmer, M.J., Benninghoff, A.D., Salinas, K.A., Williams, D.E. Proteomic screening of perfluoroalkyl acids for estrogenic activity using mass spectrometry. Toxicologist. 2010; 114: 93. |
| 177 | Tilton SC, Orner GA, Benninghoff AD, Carpenter HM, Hendricks JD, Pereira CB, Williams DE. Genomic profiling reveals an alternate mechanism for hepatic tumor promotion by perfluorooctanoic acid in rainbow trout. Environmental health perspectives. 2008 Aug;116(8):1047-55. |
| 178 | Temkin AM, Hocevar BA, Andrews DQ, Naidenko O V., Kamendulis LM. Application of the key characteristics of carcinogens to per and polyfluoroalkyl substances. Int J Environ Res Public Health. 2020;17(5):1. |